Basic Lubrication Concepts
This is a detailed overview regarding the critical properties of all motor oil lubricants. Understand the balance of additives when combined create the unique products you depend on. If anything after reading this you will know how to identify a quality synthetic motor oil and what to expect from it. I will be expanding on these topics in the near future.
Keep in mind concepts for transmission, gear and industrial synthetics are entirely a whole other beast. We are adding subsequent information on those in the particular sections.
Our main topics of discussion will include the following:
-
Friction What is it and how can it affect us
-
Lubrication – The minimization of friction
-
Basic functional demands of a required lubricant – Types or Methods of Lubrication
-
Common lubricant base stocks – Comparisons of petroleum and synthetic base stocks
-
Viscosity The number one property of a lubricant
-
Viscosity Index and Multi Grade oils
-
Additives – AMSOIL has all necessary additives. Adding an aftermarket additive is not good
Friction
Webster defines friction as the rubbing of one body against another, resistance to relative motion between two bodies in contact. Overcoming this resistance requires additional energy. Contact between the two moving surfaces generates heat and usually results in surface wear.
A simple example of friction is the heat generated when one rapidly rubs ones hands together. Note that the faster and harder you rub, the more rapid and greater the heat that is generated.Friction as a force of nature can be both beneficial and detrimental to our lives. Friction can be beneficial in several ways. As we overcome this resistance to motion between two objects in contact, heat is generated. This heat is what warms our hands or starts a fire. It is the principle behind the braking systems we find on our automobiles. Without friction, the simple task of walking would not be possible.
However, friction can also be our enemy. The heat generated as a result of friction can cause damage. Because contact is required to generate friction, wear in the areas of contact can occur. This can also lead to damage and a significant reduction in the expected life of the two bodies in contact.The reduction of friction is the primary goal behind the purpose for lubrication. Though there are many ways to reduce friction, the most common method is through the use of a fluid or semi-fluid material. When placed between two components in contact, these fluid materials will attempt to maintain a layer of separation. This occurs because such fluid materials are not readily compressible. Though the separation may be only a few millionths of an inch, it is sufficient to not only minimize contact, but in many cases, eliminated it altogether.
The inherent ability of a oil to maintain an uninterrupted film and hence component separation is referred to as lubricity. Lubricity, sometimes referred to as film strength, varies dramatically from one fluid to another.Another way to minimize friction and wear is through the use of special chemical compounds. The main role of these compounds is to offer protection when the lubricating fluid cannot maintain component separation. They may also be required to address areas of concern beyond the capabilities of the fluid itself. Examples of such areas are detergency, dispersantcy, corrosion control and acid neutralization. Chemical compounds placed in the lubricating fluid are generally referred to as additives.
Lubrication
To lubricate means nothing more than to make a surface smooth and slippery. But it can also refer to the act of apply a lubricant.
A lubricant is any substance that reduces friction by creating a slippery film between two surfaces. Lubricants permit one surface to move easily over the other surface.
Although the reduction of friction via the use of a fluid appears to be a simple concept, it is actually far more complex. Lubrication by this method can be divided into four basic types or regimes.
When a fluid lubricant is present between two rolling and/or sliding surfaces, a thicker pressurized film can be generated by the movement of the surfaces (velocities). The non-compressible nature of this film separates the surfaces resulting in no metal to metal contact. The condition in which surfaces are completely separated by a continuous film of lubricating fluid is commonly referred to as Hydrodynamic or Full Fluid Film Lubrication. In this regime, the lubricant immediately adjacent to each surface travels at the same speed and direction of each surface. As you travel through the width of the film, differential speed graduates and direction is reversed.
Hydrodynamic film thickness can be formed by wedging the lubricant through a convergent gap with the tangential surface velocities. Known as wedging film action, this is similar to a car tire hydroplaning on a wet road surface.
Although hydrodynamic lubrication is the ideal situation, in many instances it cannot be maintained. Factors which affect hydrodynamic lubrication include Lubricant Viscosity, Rotation Speed or RPM, oil supply pressure and Component Loading. An increase in speed or viscosity increases oil film thickness. An increase in load decreases oil film thickness.
Functions
Boundary Lubrication (sometimes referred to as thin film lubrication) is a condition in which the lubricant film becomes too thin to provide total separation. This may be due to excessive loading, speeds or a change in the fluids characteristics. In such a situation, contact between surface asperities (peaks and valleys) occurs. Friction reduction and wear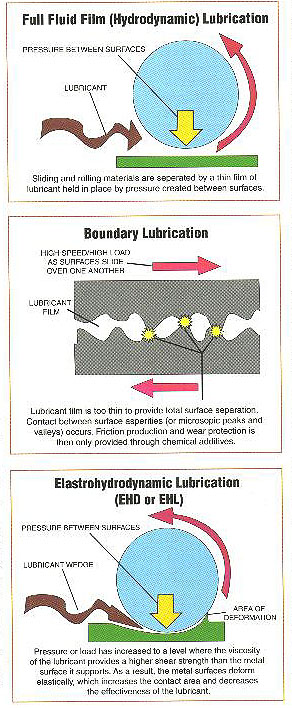 protection is then provided via chemical compounds rather than properties of the lubricating fluid. Boundary lubrication often occurs during the start up and shut down of equipment or when loading becomes excessive.
protection is then provided via chemical compounds rather than properties of the lubricating fluid. Boundary lubrication often occurs during the start up and shut down of equipment or when loading becomes excessive.
Mixed Film Lubrication is a combination of both hydrodynamic and boundary lubrication. In such a situation only occasional asperity contact occurs.
Elastohyrodynamic Lubrication (EHD or EHL) occurs as pressure or load increases to a level where the viscosity of the lubricant provides a higher shear strength than the metal surface it supports. This regime can occur in roller bearings or gears as the lubricant is carried into the convergent zone approaching a contact area or the intersection of two asperities. As a result the metal surfaces deform elastically in preference to the highly pressurized lubricant, which increases the contact area and thus increasing the effectiveness of the lubricant.
Although the ability to minimize friction and wear is the number one concern of a lubricant, there are other major functions that must be considered. A lubricant is likely to also be required to:
Clean – Maintain internal cleanliness by suspending contaminants or keeping contaminants from adhering to components.
Cool moving elements – Reducing friction will reduce the amount of heat that is generated, which will lower the operating temperature of the element or component. We also rely on the lubricant to absorb heat from the contact surface area and transport it to a location in which it can be safely dissipated.
Prevent contamination (seal) – We rely on the lubricant to act as a dynamic seal in locations such as the piston, piston ring and cylinder contact areas. This minimizes combustion by-products (for example) from contaminating the lubrication system. We also rely on the lubricant to support mechanical seals found elsewhere, minimizing external contamination and fluid loss.
Dampen shock – A lubricant may be required to cushion the blow of mechanical shock. A lubricant film can absorb and disperse these energy spikes over a broader contact area.
Prevent corrosion – A lubricant must have the ability to prevent or minimize internal component corrosion. This can be accomplished either by chemically neutralizing the corrosive products or by setting up a barrier between the components and the corrosive material.
Transfer energy – A lubricant may be required to act as an energy transfer median as in the case of hydraulic equipment or valve lifters in an automotive engine.
Base Stocks
The existence of petroleum crude has been known for hundreds of years. Yet it has only been since the 1860s that its use as a lubricant has been wide spread. One of the reasons for this is that crude, in itself, is a poor lubricant. This is due mainly to its composition. Crude is a mixture of a wide range of molecular weight hydrocarbons. Each differing molecular structure possesses a different set of properties. Though some are suitable for the production of lubricating products, many are not. Crude oil also contains high levels of contaminants such as water, sulfur, nitrogen and paraffinic material. The problem is; how can these contaminants and differing molecular structures be separated out leaving behind a more desirable stock?
The ability to separate the crude into its individual components or fractional ranges is the key. By doing this, one can obtain an improved lubricant base stock and a variety of other useful products. Distillation separates the crude into fractions based on boiling range. The crude is heated until each fraction boils off as vapor, and is then condensed and subjected to further processing. Though distillation increases the concentration of similar molecular structures, the final product still consists of a range of hydrocarbon structures. One could refer to this mixture as a chemical soup.
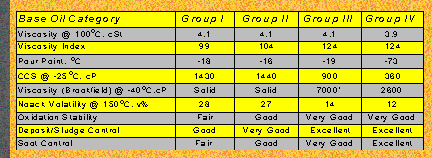
Base oil stocks can be further divided according to their level of sulfur contamination, degree of saturation and their ability to resist change in fluidity with changes in temperature. There are three categories for mineral base oils. Group I possesses the poorest set of properties and Group III offers the most desirable traits of the three. Two additional categories exist which deal with non-mineral base stocks. These are Groups IV and V. Each group is further explained on the following slide.
Oil base stocks are divided into categories based on the following physical characteristics:
Sulfur the amount of sulfur contaminate present.
Saturates The percent of molecules possessing double carbon bonds (exhibit improved properties). Unsaturated, the carbon has a single bond to a hydrogen (greater negative variation in properties).
Viscosity Index The degree to which thickness (viscosity) change occurs with changes in temperature. The higher the number the less change and the more desirable the product. Viscosity Index will be discussed in more detail later in this section.
Group I base oils are petroleum derived and the least refined of all of the groups. They are usually a mix of different hydrocarbon chains with little uniformity. While some automotive oils use these stocks, they are generally used in less demanding applications.
Group II base oils, again petroleum derived, are common in mineral based motor oils. They have fair to good performance in the areas of volatility, oxidation stability, wear prevention and flash / fire points. They have only fair performance in areas such as pour point and cold crank viscosity.
Group III base oils are subjected to the highest level of refining of all the mineral oil derived stocks. Although not chemically engineered, they offer improved performance in a wide range of areas as well as good molecular uniformity and stability. By definition, they are considered a synthesized material and can be used in the production of synthetic and semi-synthetic lubricants.
Group IV are polyalphaolefins (PAO) which are a chemically engineered synthesized base stocks. PAOs offer excellent stability, molecular uniformity and hence improved performance.
Group V base oils, with few exceptions, are also chemically engineered stocks that do not fall into any of the categories previously mentioned. Typical examples of group V stocks are Esters, polyglycols and silicone. As with group IV stocks, group V tend to offer performance advantages over groups I II. An example of a mineral based group V exception would be a white oil.

Lets look at the automotive industry. The engines used in today’s cars ask significantly more from a motor oil than they did only a few years ago. We are now using lighter oils to improve vehicle fuel economy. Yet we are asking the engines to put out more power per cubic inch of displacement then ever before. To obtain this power, we are adding turbo chargers which expose the oils to higher temperatures and greater stress. Requirements for cleaner exhaust emissions have resulted in the placement of more contamination in the oil and have again increased its operating temperature. By reducing the aerodynamic drag of new vehicles, we are minimizing the air that flows over the engine and drivetrain. Losing the cooling effect of this air flow, operating temperatures have increase even higher. A average under the hood temperature has increased from 175° F in 1975 to 225° F in 2000. Despite all of this, we are requiring the lubricants last longer than they ever did before.
Polyalphaolefins are likely to be the closest relative to mineral oil stocks of all the synthetic base materials. This is because they are both comprised of similar hydrocarbon molecules. The difference is that the Polyalphaolefin stocks consist of a single molecular structure whereas the mineral oil contains a broad range of structures. Polyalphaolefins are also known as synthesized hydrocarbons, but are most frequently referred to as PAOs. PAOs are commonly manufactured by reacting ethylene gas with a metallic catalyst. The major advantage of PAOs is their ability to function over a broader temperature range than their mineral-based counterparts.
Viscosity
Of all the characteristics a lubricant may possess, the most important is its viscosity. The viscosity of a fluid and how that viscosity reacts to variables will determine how well a fluid can perform the basic functions of a lubricant. There is no compromise for the proper viscosity!
Viscosity can be viewed in two different ways. The first is a fluids tendency to flow as it is visually seen. One can think of this as the time it takes to watch a fluid pour out of a container. The term for this is Kinematic Viscosity and it is expressed in units suggesting flow volume over a period of time. The most commonly used unit of Kinematic Viscosity is a centistoke (cSt). Another is a Sabolt Universal Second (SUS or SSU). When comparing the viscosity of two different fluids, keep in mind that a centistoke and a Sabolt Universal Second are different units, therefore they cannot be directly compared. The temperature of the fluid being tested will also affect the outcome. Kinematic viscosity of a lubricant is determined at 100°C (212°F) and/or 40°C (104°F) depending on the grading system being used. For general comparison purposes, the temperature at which the viscosity was determined for the two products must be the same.
The second is a fluids tendency to flow as is indicated by measured resistance. You can think of this as the energy required to move a object through a fluid. It takes little energy to stir water with a spoon. However, significantly more energy is required to stir honey with that same spoon. The term for this is Dynamic Viscosity or Absolute Viscosity, and is expressed in units known as centipoise (cP).
Whether it is reported in centistokes, SUS or centipoises units, the higher the number assigned, the more viscous or thicker the fluid is. The more viscous or thicker a fluid, the greater the internal friction and resistance to flow.
Fluids (lubricant stocks) thicken as they are cooled. As their temperature continues to decrease, they will eventually reach a point at which they become no longer fluid. As they thicken, their load carrying ability increases but their ability to be circulated becomes significantly impaired. As fluids are heated, they thin, which reduces their ability to prevent metal-to-metal contact. Therefore, it is important that equal temperatures be used when discussing or comparing the viscosity of fluids
Chart to be added
Viscosity Index
As previously indicated, fluids thicken as their temperature decreases and thin as they are heated. The extent that they change is indicated by their viscosity index (VI). VI is an empirical number indicating the degree of change in viscosity of an oil over a given temperature range. A high VI number indicates a relatively small change with temperature. Numbers above 95 would be considered high. A low VI number indicates a relatively large change. Numbers below 60 are considered low.
VI is based upon kinematic viscosity of a fluid tested at 40° C (104°F) and 100° C (212°F). A fluid whose viscosity changes little between these two temperatures has a higher VI than a fluid whose viscosity changes more. High VI indicates the ability to provide protection over a wider temperature range as the fluid has a greater ability to maintain its consistency.
In the chart above, the flatter the line the higher the Viscosity Index number. The greater the angle of the line, the greater the change in viscosity over temperature and the lower the Viscosity Index number.
As an example, an oil with a low VI number would tend to be very viscous at lower temperatures yet thin and very fluid at high temperature. Oils demonstrating a lesser change in viscosity will exhibit higher VI numbers.
SAE Single-grade ( sometimes referred to as straight grade or mono-grade) oils indicate their viscosity performance at either high or low temperature. They are designed for use when operating temperatures are relatively constant. Examples of high temperature single-grade motor oils would be SAE 30, 40 or 50 and SAE 80, 90 or 140 for gear lubricants. Examples of single-grade oils concerned with low temperature performance are SAE 5W, 10W or 15W for motor oils and 75W, 80W and 85W for gear lubricants. The presence of a W in a oils grade (such as 10W or 80W) indicates low temperature performance. Anytime you see the W, think of it as standing for winter or cold.
Multi-grade oils are concerned with performance at both high and low temperatures. They are designed for use when operating temperatures vary significantly. For example, if a trucker leaves Florida with a load of oranges bound for Minnesota, the trucks engine may have an SAE 30 single grade oil in it, which is appropriate for the ambient conditions in Florida. Upon reaching the sub-zero temperatures of Minnesota, the driver finds that the SAE 30 is too viscous to allow for starting of the vehicle. By draining the SAE 30 and installing an SAE 15W, he finds his starting problems are resolved and he continues his work. However, returning to the heat of Florida causes the SAE 15W to thin to the point that engine damage occurs. This could be avoided by changing back to a higher viscosity oil prior to a return to the warmer climate. The need for such frequent oil changes is not only inconvenient, it raises the cost of operation. Multi-grade oils allow for operation in changing climates without the need for changing the engine oil. Typical multi-grade engine oils are 5W-30, 10W-30, 15W-40 and 20W-50. Typical multi-grade gear lubricants are 75W-90, 80W-90 and 85W-140.
How are multi-grade (multi-viscosity) oils made? Some exist naturally. For example, many synthetic base stocks have sufficient viscosity stability (index) to qualify as multi-grade oils. For mineral base oils, it is another story. They are created by first selecting a base stock that possesses the desired low temperature properties. To that stock, chemical compounds known as viscosity index improvers are added. These viscosity index improvers minimize the thinning of the oil as temperatures increase. This allows the lighter base stock to now also meet the higher temperature requirements.
Multi-grade oils offer the best of both worlds. They provide low temperature fluidity as well as the high temperature stability required for operation in a changing environment.
When selecting a lubricant, there are three basic rules of thumb to consider.
#1 – The first rule is that the lower the starting and/or operating temperature, the lighter or thinner the selected oil should be. The higher the starting or operating temperature, the heavier or thicker the selected oil should be.
#2 – The higher the load a component will be subjected to, the heavier or thicker the oil should be. The lighter the load a component will be subjected to, the lighter or thinner the oil should be.
#3 – The faster the operating speed for a piece of equipment, the lighter or thinner the oil should be. The slower the operating speed for a piece of equipment, the heavier or thicker the oil should be.
There is no advantage in using a heavier oil than is needed. In fact, it can be a disadvantage.
Remember, too low (thin or light) = Metal-to-metal contact (friction and wear), poor sealing and increased oil consumption.
Too high (thick or heavy) = Increased fluid friction, reduced energy efficiency, higher operating temperatures and equipment starting difficulties particularly at cold temperatures.
Additives
In todays applications, base stocks alone cannot provide all the characteristics required to perform the needed functions of a lubricant. This holds true regardless of whether the stock is petroleum or synthetic in nature. However, the performance of base stocks can be enhanced by the addition of certain chemical compounds. Referred to as additives, they not only enhance existing properties, but can also impart new ones.
Additives have become an essential part of todays lubricants, as they comprise a significant amount of each quart you buy. The concentration of additives found in a typical gear lubricant for example, may range from 2-15%. As for motor oils, typical concentrations are 7.5-25%.
Oxidation Inhibitors
Oil heated in the presence of air oxidizes. As a result of this oxidation, viscosity increases as does the formation of acidic material. Deposits such as varnish and lacquer form on hot metal surfaces and may further oxidize to form sludge and hard carbonaceous materials. Oxidation increases exponentially as temperature increases. Large amounts of air (foaming for example) also increases the oxidation rate. Materials such as certain metals (copper) and acids act as catalytic agents or oxidation promoters. Although resistance to oxidation varies between different base stocks, assistance in this area is generally required. This can be accomplished through the use of Oxidation Inhibitors, which are used mainly to improve the life expectancy of a lubricant at elevated temperatures. Their use also reduces thickening of the oil and minimizes the formation of sludge and other deposits. Typical oxidation inhibitors are zinc dithiophosphates, hindered phenols, aromatic amines and sulfurized phenols.
Pour Point Depressants
In order to improve low temperature fluidity in stocks containing paraffinic (wax) materials, Pour point depressants are needed. Mineral oils produced from paraffinic crude oils are excellent examples of such stocks. As these oils are cooled, wax crystals begin to form and agglomerate into large structures. If aloud to continue, the oil becomes extremely viscous and will eventually solidify. Pour point depressants are polymers that inhibit the formation of these wax crystals and their agglomeration. Pour point depressants can only extend the lower operating temperature range of a base fluid by 5-25° F. Synthetic stocks do not contain paraffinic material and therefore do not require such additives. Their ability to function at low temperatures is a basic characteristic of their molecular structure.
Typical pour point depressants are alkylated naphthalenes and phoenolic polymers, polymethacrylates, maleata/fumarate and copolymer esters.
Viscosity Index Improvers
As we have demonstrated, a fluids viscosity will decrease as it is heated. Viscosity Index Improvers are used to reduce the thinning effects caused by operation at elevated temperatures. They are the key component that allows for the production of multi-grade oils. These oils reduce the need for oil changes due to changes in ambient temperatures.
Typical viscosity index improvers are polymers and copolymers of olefins, methacrylates, dienes or alkylated styrenes.
Viscosity Index Improvers are long chain, high molecular weight polymers that function by increasing the relative viscosity of an oil more at high temperatures than at low temperatures. Viscosity index improvers can be thought of as springs. They coil at cold temperatures and uncoil as the temperature increases. Uncoiling makes the molecules larger, which increases internal resistance within the thinning oil. This reduces the overall viscosity loss of the fluid.
The long molecules in viscosity index improvers (VII) can be subject to shearing in service, which reduces their ability to minimize fluid viscosity loss.
Permanent shear occurs when shear stress ruptures long molecules and converts them into shorter and lower weight molecules. The shortened molecules offer less resistance to flow and minimizes their ability to maintain viscosity. Permanent shearing of viscosity index improvers can result in piston ring sticking (due to deposit formation), increased oil consumption and accelerated equipment wear. It should be noted that some VIIs are significantly more shear stable than others. Also, although the type of base stock used and the intended application determines the need for VII, many synthetic stocks may not require them at all.
Loss of fluid viscosity can also occur due to a condition known as Temporary Shear. Temporary shear occurs when long viscosity index improver molecules align themselves in the direction of the stress (flow). This alignment generates less resistance and allows for a reduction in fluid viscosity. When the stress is removed, the molecules return to their random arrangement and the temporary loss in viscosity is recovered.
Anti-wear (AW) and extreme pressure (EP) additives
To provide wear protection when the oil film alone is not capable of preventing contact between components, Anti-wear (AW) and extreme pressure (EP) additives must be used. This is typically the case in boundary lubrication. These additives work by providing a sacrificial wear surface or by changing the surface metallurgy of the components.
Anti-wear additives or their reaction products form thin, tenacious films on loaded parts to prevent metal-to-metal contact. These additives assist in the reduction of friction, wear, scuffing and scoring under boundary lubrication conditions. Typical anti-wear additives include zinc dithiophosphate and polar molecules such as fatty oils, acids and esters.
Extreme pressure additives are also commonly referred to as EP additives (these are not needed with typical synthetic engine oils). Like anti-wear additives, they or their reaction products also form thin, tenacious films but on heavily loaded or shock loaded components. Sulfur containing compounds are commonly used as EP additives. The sulfur forms a sacrificial film on components so contact occurs between the sulfurized films instead of the components themselves. Certain chlorinated compounds may also be used as EP additives. However, current use of chlorinated compounds is limited due to environmental and corrosion concerns.
Rust and corrosion inhibitors
Additives that form protective films on metal surfaces, protecting them against attack by water and/or corrosive contaminants are necessary but seldom incorporated in most engine oils. Rust and corrosion inhibitors additives are needed as oil by itself tends to offer very little protection. Corrosion of an acidic nature is handled through acid neutralization. Sulfanated compounds are the most widely used additives for this purpose. Generally these additives are not inherent in engine oils and can in fact work in opposition to lubrication properties. AMSOIL has introduced technologies recently not used by our competition and is specifically beneficial in our motorcycle oils.
Detergents
One of the most important aspects to a quality synthetic engine oil are detergents. These are added to lubricants to minimize deposit formation in the high temperature areas of an engine. The most commonly used detergents in motor oil formulations are metallic (ash) soaps with reserve basicity to neutralize acids formed as a by-product of combustion.
Detergents are generally used in conjunction with dispersants. Common detergents are metallo-organic compounds of sodium, calcium and magnesium phenolates, phosphonates and sulfonates.
AMSOIL synthetic motor oils contain detergents designed to last beyond advertised basic PH levels in other competitor’s products.
Dispersants
Additives that help keep solid contaminants in suspension within a lubricant to prevent sludge, varnish and other carbon deposits from forming on engine partsare dispersants. They also prevent contaminants from agglomerating into larger, potentially dangerous particles. Typical dispersants are alkysuccinmides, alkylsuccinic esters and mannich reaction products.
Friction Modifiers
Additives that alter the coefficient of friction of a lubricant are referred to as friction modifiers. Organic fatty acid and amides, lard oil and high molecular weight organic phosphoric acid esters are all common friction modifiers. Friction modifiers of this nature are commonly used in automotive engine oils to improve fuel economy. Other friction modifiers are suspended solids such as PTFE (TEFLON), graphite and molybdenum (moly).
Anti-Foaming Agents
When a fluid is agitated, air can be trapped forming bubbles. This is commonly referred to as foaming. Because air is compressible, the ability of the fluid film to prevent contact is reduced. And because the mixed air contains oxygen, oxidation of the fluid is promoted.
Although it is difficult to prevent this process from occurring, measures can be taken to minimize its effect. One way is through the use of ANTI-FOAMING agents. For example, silicone compounds can be used to reduce the surface tension of the air bubbles, which will result in a more rapid breakdown of surface bubbles. However, care must be taken, as excess amounts of these agents will promote foaming. Organic copolymers may be used to decrease the number of small entrained bubbles.
Foaming can also be caused by air leaks within the vacuum side of a system. The presence of water and surface active materials such as rust preventatives, detergents and solvents can also promote foaming.
Today’s additive systems can be quite sophisticated. They can be chemically sensitive and negatively affected by the addition of other chemicals. Therefore, you never want to intentionally mix lubricants of different brands or types.
Though there may be a variety of additives directed toward a specific function, the performance of each can differ significantly. The concentration at which an additive is used will also have a major impact on how well a lubricant performs a given task.
Although the use of additives is generally advantageous, their presence in a lubricant can result in negative side effects. For example, detergents and dispersants can also promote foaming and minimize the effectiveness of anti-foaming additives. Certain additives, EP for example, can promote corrosion of copper, bronze or brass containing components when exposed to high temperatures. EP additives can also sacrifice thermal stability of the base oil.
In the above graph, we see that the presence of an extreme pressure additive actually increases friction at lower loads. It would be counterproductive to utilize an EP additive in an application in which loading is not adequate to take advantage of the additive used. Another consideration is that EP additives tend to produce corrosive by-products when subjected to elevated temperatures. The rule of thumb here is that if an application does not require an additive do not use it. This holds true especially for aftermarket additives.
Look and consider the total package. Only when an oils base stock and additive package are working in unison will it be capable of providing the required basic functions of:
Reducing friction and wear
Clean
Cooling moving elements
Preventing contamination (seal)
Dampen shock
Transfer energy
Prevent corrosion
Never consider utilizing a lubricant based solely on its performance in a single area. Lubricants must perform well in all areas in order to be suitable candidates.
Archive Source – Now totally updated in 2018 with the new AMSOIL University –
AMSOIL Lubrication I training manual (note: This is only part of a complete training program designed for AMSOIL dealers. The purchase of these modules are highly recommended and upon passing the online examinations for the entire series, AMSOIL rewards dealers with credits for clothing and literature items. Modules Available:
Lubrication 1&2
Engine Oils
Mobile Drive Train Fluids
Filtration
Oil Analysis
Fuel Additives and grease
Join our bandwagon and create a back-up income for your self by becoming an AMSOIL Dealer today. Either call us or look over the AMSOIL Dealer signup information. It’s quite the opportunity! AMSOIL now hosts yearly the AMSOIL University. Dealers attending have an unprecedented edge once knowledgeable of the basic concepts and opportunities they have at hand.
